If you read my recent article on sparging session beers, you’ll know that fully sparging the grain bed for a low-gravity session beer can yield less volume than a typical pre-boil wort target. For a 5.0-gallon (19-L) batch of session beer, you’ll collect less than 6.0 gallons (23 L) of wort and perhaps even less than 5.0 gallons (19 L). The simplest way to deal with this is to add water to your wort to yield a volume you can boil for 60 to 90 minutes. Another way to deal with it would be to boil the smaller volume for your desired boil time, then add cool water to the post-boil wort in the kettle. This way saves you a little time when chilling the wort.
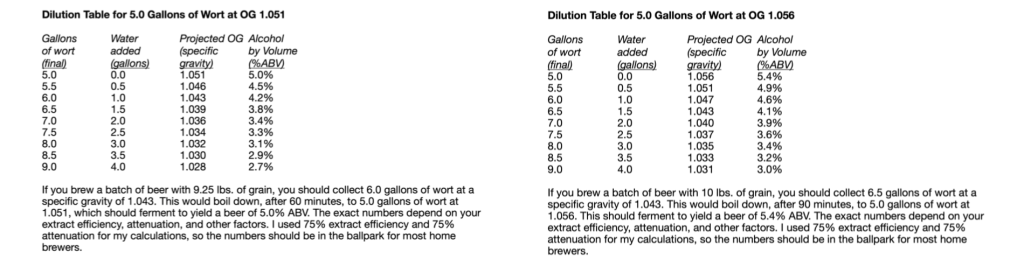
However, there is a third possibility — one that will yield more beer with the same amount of effort and using your same equipment (except perhaps for a second fermenter). The third possibility is to select an amount of grain that will yield either 6.0 gallons (23 L) or 6.5 gallons (25 L) of wort from a fully-sparged grain bed. This is 9.25 lb. (4.2 kg) and 10.0 lb. (4.5 kg), respectively. This will allow you to perform a 60-minute boil or a 90-minute boil, whichever you prefer. In either case, your pre-boil wort should have a specific gravity (SG) around 1.043. After a 60-minute boil of the 6.0 gallons (23 L), your original gravity (OG) should be close to 1.051. For a 90-minute boil of the 6.5 gallons (25 L), 1.056. Then, dilute this wort in your fermenter to your target original gravity. (In practice, your numbers may vary slightly from these. If you try this, take good notes on your grain weight, your wort yield, and the density of your pre-boil and post-boil wort. With that information, you can tweak your process, if needed, the next time you brew.)
The two tables above show you how much dilution water you need to hit a variety of original gravities in the session beer range. As an example, let’s say you wanted to brew an ordinary bitter at an OG of 1.036. Furthermore, you wanted to start with 9.25 lb. (4.2 kg) of grain and boil for 60 minutes. On the appropriate table, find the OG of 1.036. Look to the left of that number to see you’ll have to add 2.0 gallons (7.8 L) of water to the 5.0 gallons of post-boil wort for a yield of 7.0 gallons (26 L) at SG 1.036. Once you know this, formulate a recipe for 7.0 gallons (26 L) of bitter, but brew it as a 5.0-gallon (19-L batch). After the boil, chill the wort and dilute it to your target strength in your fermenter (or fermenters).
Using this method, you can make more than 5.0 gallons (19 L) of beer with the almost the same amount of effort as brewing a 5.0-gallon (19-L) batch. And you won’t need a larger mash tun or kettle. All you’ll need is more fermenter space. Since session beers are meant to be “the one to have when you’re having more than one,” it can be good to brew a little extra.




















Recent Comments