This is another installment in the Enzymes for Brewers series.
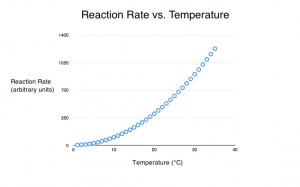
The rate of an enzymatic reaction increases with temperature. This is because the molecules in solution are traveling at a higher speed, lowering the average time it takes for them to collide. Back in 1889, the Dutch chemist Svante Arrhenius derived a simple equation that showed the effect of temperature on the rate of any chemical reaction. Basically, his equation predicts that the rate of any chemical reaction will increase exponentially with temperature. Experiments have shown that his equation is remarkably accurate for a wide range of chemical reactions.
In chemistry, a rough rule of thumb is that, for every 10 °C increase in temperature, the rate of a reaction doubles. This is only an approximation, and the exact rate of increase depends on a rate constant specific to each reaction. (A rate constant is just a number that describes how fast the reaction proceeds. It has to be measured for each specific reaction.) However, for a surprisingly wide number of “simple” chemical reactions, this rule of thumb is “close enough” for most practical purposes. If the reaction is actually a net reaction of a series of chemical reactions, or a cascading reaction, this approximation can be way off.
So, the rate of a chemical reaction increases at higher temperatures. However, at high temperatures, proteins can denature. When an enzyme is denatured, it loses its shape. Basically, the string of amino acids that were folded into a 3-D “ball” are now unraveled because the molecule is “shook apart” by the heat. As such, there is no active site, and the enzyme cannot bind with the substrate to form the enzyme-substrate complex. In solution, the progressive denaturation of enzymes means that rate of reaction eventually drops to zero. Changes in pH and salt concentration can also denature proteins.
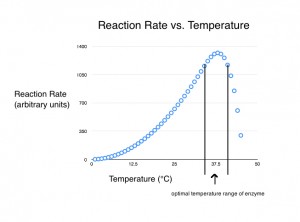
The denaturation of proteins does not happen instantly. When a solution is heated above an enzyme’s denaturation point, the number of intact enzymes begins to decrease. The rate of inactivation varies among enzymes. At progressively higher temperatures, the rate of enzyme destruction increases. For the shrinking pool of surviving molecules, the reaction rate is still increasing with temperature. But this is offset — and eventually overwhelmed — by the growing pool of denatured molecules.
A Simple Model
So, a simple model of how enzyme performance is affected by temperature would start at low temperatures and include a range over which increases in temperature caused exponential increases in the rate of reaction. Above this, there would be a range of temperatures in which the increase in the rate of reaction was offset by the denaturation of the enzymes. In this range, the initial rate of reaction would be higher than at cooler temperatures, because only a small percentage of the enzyme molecules would have denatured. However, over time the reaction rate would decrease as more of the molecules were destroyed. And finally, there would be a range of temperatures at which all the enzyme molecules were denatured and the rate of reaction was zero.
In this model, enzymes are not “switched on” when the mash reaches a certain temperature. Indeed, there is nothing resembling a temperature-dependent switch on the enzymes relevant to brewing (or any other enzyme that I have heard of). They are just a big “blob” floating in solution. Below the “active range” of brewing enzymes, the activity of the enzyme is increasing along a steady curve. Likewise, enzymes are not instantly “switched off” over the high-end of the optimal range. Indeed, the high-end of the optimal range for many brewing enzymes is at an arbitrary point at which activity is a trade-off between heat-stimulated activity and denaturation.
In the brewing literature, an optimal temperature range is often given for certain enzymes. The low end of this range is the point at which the enzyme will complete its task in a reasonable time. The high end is the point at which sufficient enzyme activity will remain to finish the job, given a reasonable mash duration. On occasion, a single temperature will be given as the optimum for an enzyme for a particular task. Likewise, a temperature at which the enzyme denatures may also be given. Sometimes this is the temperature at which the enzyme begins to denature, other times it is when the process of denaturation accelerates greatly, relevant to reasonable brewing timeframes.
In brewing, the optimum temperature for an enzyme depends on the pH of the mash, mash thickness, and the concentration certain ions (for example, alpha amylase is stabilized by the presence of calcium ions). Temperatures given usually assume “normal” mash conditions. No single optimum temperature or temperature should be considered “the” correct answer.
In the next article, which I’ll post tomorrow, we’ll look at the kinetics of enzymes — as best they are known — in the mash.
—
Related articles

Speak Your Mind