 Recently, I posted four simple water guides. They described a simple way to make up your brewing water (brewing liquor) for pale, amber, brown, and black beers. Each gave a recipe for adding minerals to 5.0 gallons (19 L) of distilled water to make water suitable for beers in the appropriate color range. Three of the four guides were based on starting with 100 ppm calcium ions (Ca2+) in your water; the fourth started with a base of 75 ppm.
Recently, I posted four simple water guides. They described a simple way to make up your brewing water (brewing liquor) for pale, amber, brown, and black beers. Each gave a recipe for adding minerals to 5.0 gallons (19 L) of distilled water to make water suitable for beers in the appropriate color range. Three of the four guides were based on starting with 100 ppm calcium ions (Ca2+) in your water; the fourth started with a base of 75 ppm.
There’s nothing magical about 100 ppm Ca2+, however. It is a middle value in the range of useful calcium levels in beer, often given as 50–200 ppm. Calcium has a variety of benefits in the mash, and brewing liquor in this range should supply a sufficient amount. If you are making your brewing liquor from distilled water, the more calcium you add, the more carbonates you need to add. Calcium causes a reaction in the mash that releases acids and lowers pH. Carbonates neutralize acid and oppose mash acidification. So, to hit your proper mash pH, you need these two to be (at least somewhat) in balance. With that in mind, I’ve made a five-step guide to making your brewing liquor that allows you to start with 50, 75, 100, or 150 ppm calcium ions. Beers brewed from water containing more overall minerals may taste “minerally” while beers brewed with water containing lower levels of minerals may taste “softer” or “more rounded.”
Five Step Easy Water Guide
Start with 5.0 gallons (19 L) of distilled water. (For 5.0-gallon (19-L) all-grain batches, you will need to make more than 5.0 gallons (19 L) of brewing liquor. Scale the recipe up appropriately. For extract batches, you may only need to make half of this volume if you use distilled water for the dilution water.) Then, just follow the steps to make your water. Be sure to taste it before brewing.
Step 0.) Chloramines
You could also use tap water with a very low mineral content (total dissolved solids under 25 ppm) as a starting point. However, if you do so, you will need to eliminate the chloramines in the water first. Either filter through a large carbon filter or add one Campden tablet per 20 gallons (76 L).
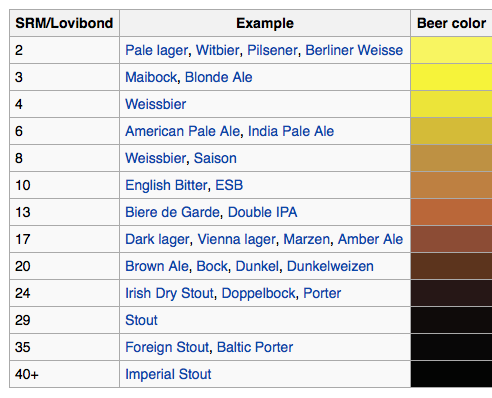
Step 1.) Estimate Your Beer’s SRM
Calculate the color of your beer, based on the grain bill, in SRM. If your recipe contains darkly roasted grains (>200 °L), recalculate this number following the instructions in this article.
Step 2.) Calcium
Choose the level of calcium you wish to start with. For most beers, anything in the 50–200 ppm range is fine. Also choose whether your beer is going to be balanced or if you’d like the hop bitterness to be accentuated. Anything over 1.75 tsp. gypsum per 5.0 gallons (19 L) will yield brewing liquor with at least 200 ppm sulfate; this is considered best for very hoppy beers. Add the specified amount of minerals to your 5.0 gallons (19 L) of brewing liquor.
Step 3.) Carbonate
Locate the approximate color of your beer (in SRM) from the leftmost column, then chose the amount of baking soda (NaHCO3) to add to your 5.0 gallons of calcified brewing water from the appropriate column to the right. (If you want to know the concentration of carbonates in your water, 1 tsp in 5.0 gallons adds 170 ppm carbonate — sort of. The actual amount of pH dependent, but don’t worry about that for now.)
Any addition of baking soda over 1.6 tsp. per 5.0 gallons (19 L) will lead to sodium levels being over 100 ppm. This may not be healthy for people who are on a sodium restricted diet. Any addition of baking soda over 2.4 tsp. per 5.0 gallons (19 L) will lead to sodium levels being over 150 ppm and likely add a salty taste. This is definitely not recommended.
Step 4.) Magnesium (Optional)
Brewing liquor may contain up to 40 ppm magnesium sulfate (MgSO4). You can add magnesium or not, it’s your choice. Some brewers feel that around 30 ppm benefits dark beers. Magnesium has a small effect on mash pH, but not so great that adding the amounts here would throw your mash chemistry off.
Step 5.) Add Calcium to the Boil, If Needed
Making your water as described above should enable your mash to fall in the proper pH range — 5.2–5.6. Optimally, your pH should drop to 5.0–5.2 near the end of the boil. Sometimes, a little calcium is needed to accomplish this. When you boil, look at to your hot break. Once formed, it should be big and fluffy. If not, add about 50 ppm calcium to the boil. For 5.0 gallons (19 L), this is a teaspoon of either calcium chloride or gypsum. If this doesn’t help, try adding another 50 ppm calcium.
Using this simple guide should allow you to get your water chemistry into the right ballpark for a proper mash pH to establish itself. Later, you should be set up to hit the proper post-boil pH, too. (A bit of added calcium may be required to bring this about). Matching your brewing liquor to the grain bill of your beer should result in better beer. Take good notes and, if possible, compare batches using untreated and treated water to see if adjusting your water chemistry is improving your beer.
If you enjoy Beer and Wine Journal, please consider supporting us by clicking on one of the donate buttons, thanks.
—
Related Article




I love this! So simple, but so perfect at the same time. Since my water is usually an odd volume it may be easier to weigh my salt additions. how many grams per tsp did you use for gyp. Cacl, & baking soda? Also, I do biab so I mash full volume. Do you think I should use less baking soda than you call for to counteract the effect of a more dilute mash wanting to raise the mash ph? Thanks
How much a tsp. of each of these minerals weighs depends on how tightly they are packed, but I usually go with:
1 tsp gypsum = 4.0 grams
1 tsp calcium chloride = 3.4 grams
1 tsp sodium bicarbonate = 4.4 grams
And no, I would nt use less baking soda. I’d keep the same concentration of minerals regardless of mash thickness.
Stupid question but you mention distilled water, and I use RO water. These are not exactly the same but I assume it doesn’t change the salt additions???
Thanks for a great chart and explanation.
RO water should work as well as distilled. Both should have close to zero dissolved solids to start with.
I absolutely love this — after a year of messing around with brew’n water with some frustration I finally found this which will be my new method! This is the easy water solution that I was looking for all along. I’ve already shared with several brewing friends, thanks!!!
Thanks for this guide. This is very informative and very easy to understand. It’s simple. I appreciate blogs like this.
As always, this time you also successfully stopped me to appreciate all of your work. you always come up with the great informational article. I would like to thank you for caring your audience
I’m a Newby to all this and I’m looking for the most basic solution to water chemistry. But I have no idea what ppm CA ++ is can anyone elaborate please
What about sulfates and chlorides, and the appropriate ratio for a given style?
Just wanted to say thanks for this. I’ve always agreed that water chem is a giant part of honing in on great beer but everything I’ve ever read assumes we are chemists and I give up on it. This article is so simple and easy to understand. I truly appreciate it.
hey. this is used for industrials and others. i read this in irchem.ir